Detecting and zapping Alzheimer’s and Parkinson’s with lasers
November 20, 2013
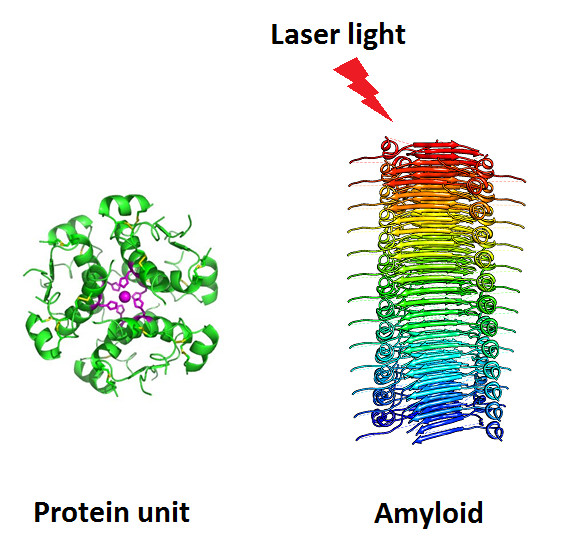
Researchers
at Chalmers University of Technology in Sweden, together with
researchers at the Polish Wroclaw University of Technology, have made a
discovery that may lead to curing diseases such as Alzheimer’s,
Parkinson’s and Creutzfeldt-Jakob disease with phototherapy (credit:
Chalmers University of Technology)
Today, amyloid protein aggregates are treated with highly toxic chemicals for detection and removal.
With a multiphoton laser, the chemical treatment would be unnecessary, and it might be possible to remove the harmful protein without touching the surrounding tissue, the researchers suggest.
These diseases arise when amyloid beta protein are aggregated in large doses so they start to inhibit proper cellular processes.
Different proteins create different kinds of amyloids, but they generally have the same structure. This makes them different from the well-functioning proteins in the body, which can now be shown by multi photon laser technique.
Abstract of Nature Photonics paper
Fibrillization of peptides leads to the formation of amyloid fibres, which, when in large aggregates, are responsible for diseases such as Alzheimer’s and Parkinson’s. Here, we show that amyloids have strong nonlinear optical absorption, which is not present in native non-fibrillized protein. Z-scan and pump–probe experiments indicate that insulin and lysozyme β-amyloids, as well as α-synuclein fibres, exhibit either two-photon, three-photon or higher multiphoton absorption processes, depending on the wavelength of light. We propose that the enhanced multiphoton absorption is due to a cooperative mechanism involving through-space dipolar coupling between excited states of aromatic amino acids densely packed in the fibrous structures. This finding will provide the opportunity to develop nonlinear optical techniques to detect and study amyloid structures and also suggests that new protein-based materials with sizable multiphoton absorption could be designed for specific applications in nanotechnology, photonics and optoelectronics.
(¯`*• Global Source and/or more resources at http://goo.gl/zvSV7 │ www.Future-Observatory.blogspot.com and on LinkeIn Group's "Becoming Aware of the Futures" at http://goo.gl/8qKBbK │ @SciCzar │ Point of Contact: www.linkedin.com/in/AndresAgostini
 Washington
Washington