Biomaterial-delivered chemotherapy could effectively treat brain tumors
November 20, 2013
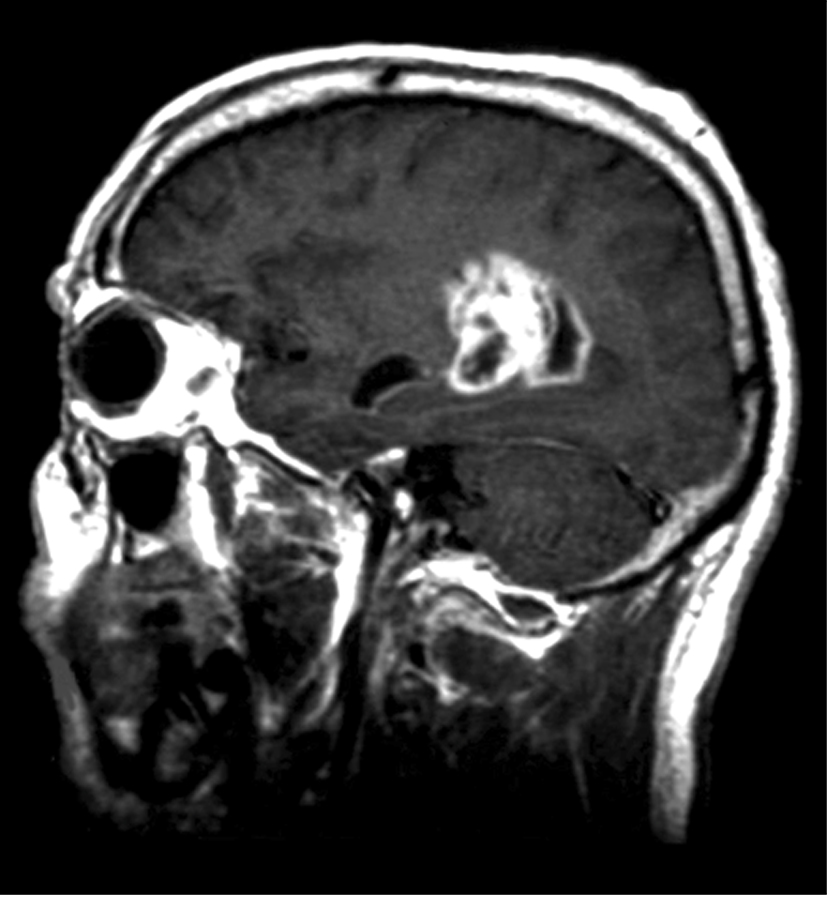
An
image of a sagittal section MRI scan of a patient with glioblastoma
multiforme, one of the brain tumor types most amenable for a new drug
delivery system (credit: University of Nottingham)
Their study, published in the journal PLOS ONE (open access), shows that the biomaterial can be easily applied to the cavity created following brain cancer surgery and used to release chemotherapy drugs over several weeks.
The targeted nature of the therapy could also reduce the toxic effects of chemotherapy drugs on healthy parts of the body, potentially reducing the debilitating side-effects that many patients experience after cancer treatment.
Patient survival
“Our system is an innovative method of drug delivery for the treatment of brain tumors and is intended to be administered immediately after surgery by the operating neurosurgeon,” said Dr. Ruman Rahman, of the University of Nottingham’s Children’s Brain Tumour Research Centre (CBTRC), who led the study.
“Ultimately, this method of drug delivery, in combination with existing therapies, may result in more effective treatment of brain tumors, prolonged patient survival and reduced morbidity.”
“Although alternative systems for delivery of drugs directly to the brain have been developed, they are used infrequently because their success has been limited,” Rahman told KurzweilAI. “This new drug delivery system is the first that can be molded to the shape of the brain tumor cavity and the first to deliver several different drugs over a clinically meaningful period of time.”

A
zoomed in electron microscope image of the structure of the polymer
once it has solidified. At this magnified resolution, what you can see
are the individual microparticles (credit: University of Nottingham)
The Nottingham polymer formulation is made from two types of microparticles called PLGA and PEG. A powder at room temperature, it can be mixed to a toothpaste-like consistency with the addition of water.
The unique properties of the polymer lie in its ability to set into a rigid structure only when it reaches body temperature, a feature perfectly tailored for use in medical therapies. It was originally developed as a scaffold onto which new bone cells could be grown to speed up the knitting back together of broken bones.
The cavity left by the removal of a tumor would be lined with the polymer while in paste form, which would start to solidify and gradually release the chemotherapy drugs after the incision has been closed. This would directly target any residual cells not initially removed during surgery.
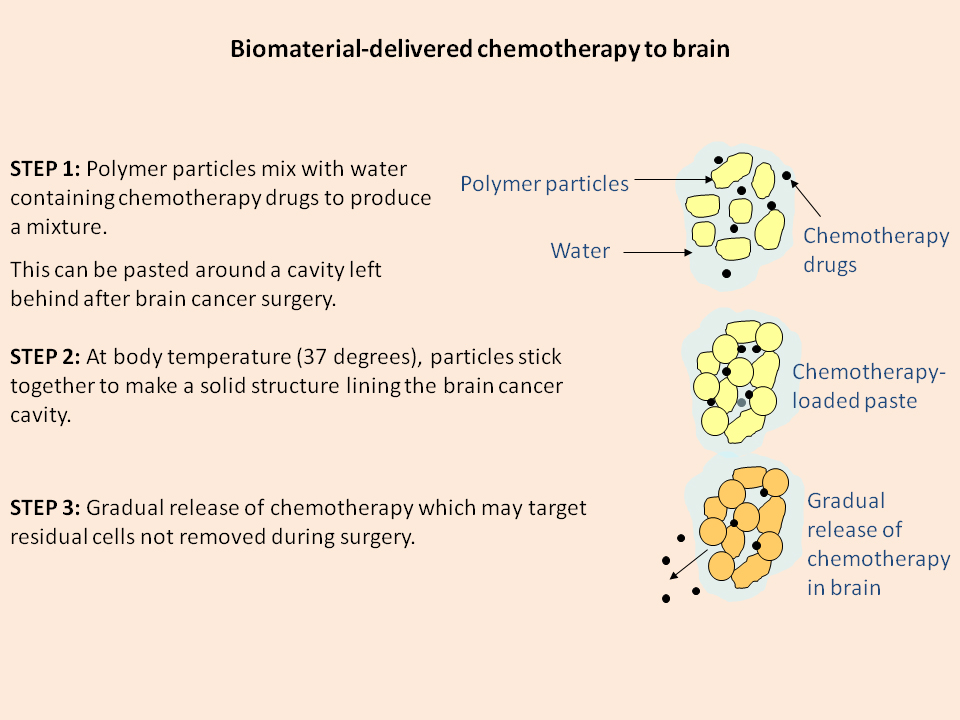
A cartoon depicting how the polymer-based chemotherapy system works (credit: University of Nottingham)
To establish whether the material itself is safe to use on patients in this form of therapy, they used it to create a 3D model onto which they were able to grow brain tumor cells and healthy brain blood vessel cells without any toxicity. They then simulated surgery on a sheep’s brain from an abattoir by molding the paste around a brain cavity and warming the brain to human body temperature to harden the polymer.
The brain was then scanned using CT and MRI technology to demonstrate that it is still possible to distinguish the polymer from normal brain tissue on a routine brain scan, an aspect crucial for doctors when dealing with follow-up care for brain tumor patients who have undergone surgery.
Robust material
The team also dealt with concerns that the material could disintegrate and release its chemotherapy contents too quickly during the subsequent radiotherapy which many cancer patients undergo following surgery. By placing the biomaterial loaded with chemotherapy drugs into a head cavity of a medical training dummy and subjecting it to the same duration and intensity of radiotherapy used for brain tumor patients they were able to successfully demonstrate the robust integrity of the structure.
Finally they showed that a chemotherapy drug called etoposide could be effective at killing brain cancer cells in a mouse when released from the polymer formulation.
The next stage of the research will be to extend the study in mice with brain tumors to test whether animals with the drug-loaded polymers survive longer. The team is also investigating the release of other chemotherapeutic drugs that hold promise, supported by a recent grant award from Sparks.
As the research used a biomaterial and chemotherapy drugs already approved for medical use, many of the usual ethical approval hurdles to allow further investigation have already been cleared.
The first clinical test, anticipated in 3 years’ time, will be to devise a clinical trial that would involve testing the therapy on a small number of patients for whom other clinical treatments have not been successful and would otherwise only be offered palliative care. “If successful, a commercially available product could be available in 5–7 years time,” Rahman said.
The study was funded by a grant from the Joseph Foote Trust, now part of the Brain Tumour Charity and a Nottingham Advanced Research Fellowship from The University of Nottingham.
Abstract of PLOS ONE paper
Introduction. Polymer-based delivery systems offer innovative intra-cavity administration of drugs, with the potential to better target micro-deposits of cancer cells in brain parenchyma beyond the resected cavity. Here we evaluate clinical utility, toxicity and sustained drug release capability of a novel formulation of poly(lactic-co-glycolic acid) (PLGA)/poly(ethylene glycol) (PEG) microparticles.
Methods. PLGA/PEG microparticle-based matrices were molded around an ex vivo brain pseudo-resection cavity and analyzed using magnetic resonance imaging and computerized tomography. In vitro toxicity of the polymer was assessed using tumor and endothelial cells and drug release from trichostatin A-, etoposide- and methotrexate-loaded matrices was determined. To verify activity of released agents, tumor cells were seeded onto drug-loaded matrices and viability assessed.
Results. PLGA/PEG matrices can be molded around a pseudo-resection cavity wall with no polymer-related artifact on clinical scans. The polymer withstands fractionated radiotherapy, with no disruption of microparticle structure. No toxicity was evident when tumor or endothelial cells were grown on control matrices in vitro. Trichostatin A, etoposide and methotrexate were released from the matrices over a 3-4 week period in vitro and etoposide released over 3 days in vivo, with released agents retaining cytotoxic capabilities. PLGA/PEG microparticle-based matrices molded around a resection cavity wall are distinguishable in clinical scanning modalities. Matrices are non-toxic in vitro suggesting good biocompatibility in vivo. Active trichostatin A, etoposide and methotrexate can be incorporated and released gradually from matrices, with radiotherapy unlikely to interfere with release.
Conclusion. The PLGA/PEG delivery system offers an innovative intra-cavity approach to administer chemotherapeutics for improved local control of malignant brain tumors.
(¯`*• Global Source and/or more resources at http://goo.gl/zvSV7 │ www.Future-Observatory.blogspot.com and on LinkeIn Group's "Becoming Aware of the Futures" at http://goo.gl/8qKBbK │ @SciCzar │ Point of Contact: www.linkedin.com/in/AndresAgostini
 Washington
Washington