Creating living brain cells from deceased Alzheimer’s patients biobanked brain tissue
New stem cell lines will allow researchers to “turn back the
clock” and observe how Alzheimer’s develops in the brain, potentially
revealing the onset of the disease at a cellular level
January 9, 2014
[+]
Scientists at The New York Stem Cell Foundation
(NYSCF) Research Institute, working in collaboration with scientists
from Columbia University Medical Center (CUMC), have for the first time
generated induced pluripotent stem (iPS) cells lines from
non-cryoprotected brain tissue of patients with Alzheimer’s disease.
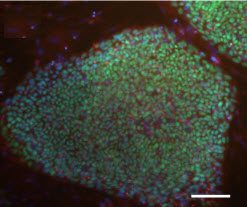
Staining
of these biobank IPS cells show they retain pluripotency (scale bar:
100 microns)(credit: Andrew A. Sproul et al./Acta Neuropathologica
Communications)
These new stem cell lines will allow researchers to “turn back the clock” and observe how Alzheimer’s develops in the brain, potentially revealing the onset of the disease at a cellular level long before any symptoms associated with Alzheimer’s are displayed. These reconstituted Alzheimer’s cells will also provide a platform for drug testing on cells from patients that were definitively diagnosed with the disease.
Until now, the only available method to definitively diagnose Alzheimer’s disease that has been available to researchers is examining the brain of deceased patients. This discovery will permit scientists for the first time to compare “live” brain cells from Alzheimer’s patients to the brain cells of non-Alzheimer’s patients.
The scientists successfully produced the iPSCs (induced pluripotent stem cells) from frozen tissue samples stored for up to eleven years at the New York Brain Bank at Columbia University.
This advance, published (open access) in Acta Neuropathologica Communications, shows that disease specific iPS cells can be generated from readily available biobanked tissue that has not been cryoprotected, even after they have been frozen for many years. This allows for the generation of iPS cells from brains with confirmed disease pathology and allows access to rare patient variants that have been banked.
In addition, findings made using iPS cellular models can be cross-validated in the original brain tissue used to generate the cells. The stem cell lines generated for this study included samples from patients with confirmed Alzheimer’s disease and four other neurodegenerative diseases.
First living diagnosis of Alzheimer’s and first use of existing biobanks
This important advance opens up critical new avenues of research to study cells affected by disease from patients with definitive diagnoses. This success will leverage existing biobanks to support research in a powerful new way.
iPS cells are typically generated from a skin or blood sample of a patient by turning back the clock of adult cells into pluripotent stem cells, cells that can become any cell type in the body. While valuable, iPS cells are often generated from patients without a clear diagnosis of disease and many neurodegenerative diseases, such as Alzheimer’s disease, often lack specific and robust disease classification and severity grading.
These diseases and their extent have only been definitively diagnosed by post-mortem brain examinations. For the first time we will now be able to compare cells from living people to cells of patients with definitive diagnoses generated from their banked brain tissue.
Brain bank networks, which combined contain tens of thousands of samples, provide a large and immediate source of tissue, including rare disease samples and a conclusive spectrum of disease severity among samples. The challenge to this approach is that the majority of biobanked brain tissue was not meant for growing live cells, and thus was not frozen in the presence of cryoprotectants normally used to protect cells while frozen.
However, NYSCF scientists in collaboration with CUMC scientists have shown that these thousands of samples can now be used to make living human cells for use in disease studies and to develop new drugs or preventative treatments for future patients.
The New York Stem Cell Foundation research was supported by the Cure Alzheimer’s Fund, Charles Evans Foundation, and The New York Stem Cell Foundation. The Columbia University Medical Center research was funded by the Alzheimer’s Association, the Louis V. Gerstner, Jr., Foundation, and The American Recovery and Reinvestment Act (ARRA).
New gene targets in early-onset Alzheimer’s
NYSCF scientists have also successfully generated a stem cell model of familial Alzheimer’s disease (FAD), working with scientists at the Icahn School of Medicine at Mount Sinai (ISMMS). Using this stem cell model, researchers identified fourteen genes that may be implicated in the disease and one gene in particular that shows the importance that inflammation may play in the brain of Alzheimer’s patients.
In this study, published (open access) in PLOS ONE, the team of scientists produced stem cells and neural precursor cells (NPCs), representing early neural progenitor cells that build the brain, from patients with severe early-onset AD with mutations in the Presenilin 1 (PSEN1) gene.
These NPCs had elevated Abeta42/Abeta40 ratios, indicating elevation of the form of amyloid found in the brains of Alzheimer’s patients. These levels were greater than those in adult cells that did not have the PSEN1mutation. This elevated ratio showed that these NPCs grown in the petri dish were accurately reflecting the cells in the brains of FAD patients.
“Our ability to accurately recapitulate the disease in the petri dish is an important advance for this disease. These genes provide us with new targets to help elucidate the cause of sporadic forms of the disease as well as provide targets for the discovery of new drugs,” said Susan L. Solomon, Chief Executive Officer of The New York Stem Cell Foundation.
“The gene expression profile from Noggle’s familial Alzheimer’s stem cells points to inflammation, which is especially exciting because we would not usually associate inflammation with this particular Alzheimer’s gene,” said Sam Gandy, MD, PhD, Professor of Neurology and Psychiatry and Director of the Center for Cognitive Health at the Icahn School of Medicine at Mount Sinai and a co-author on the study. Gandy is also Associate Director of the NIH Designated Mount Sinai Alzheimer’s Disease Research Center.
The researchers generated induced pluripotent stem (iPS) cells from affected and unaffected individuals from two families carrying PSEN1 mutations. After thorough characterization of the NPCs through gene expression profiling and other methods, they identified fourteen genes that behaved differently in PSEN1 NPCs relative to NPCs from individuals without the mutation. Five of these targets also showed differential expression in late onset Alzheimer’s disease patients’ brains. So in the PSEN1 iPS cell model, the researchers reconstituted an essential feature in the molecular development of familial Alzheimer’s disease.
Although the majority of Alzheimer’s disease cases are late onset and likely result from a mixture of genetic predisposition and environmental factors, there are genetic forms of the disease that affect patients at much earlier ages. PSEN1 mutations cause the most common form of inherited familial Alzheimer’s disease and are one hundred percent penetrant, resulting in all individuals with this mutation getting the disease.
The identification of genes that behaved differently in patients with the mutation provides new targets to further study and better understand their effects on the development of Alzheimer’s disease. One of these genes, NLRP2, is traditionally thought of as an inflammatory gene.
“The fact that the NLRP2 gene is upregulated in these cells is interesting because inflammatory genes have long been implicated in late onset and sporadic forms of the disease. The importance is still unknown, but these cells may provide a platform to understand the function of this gene as well as others that contribute to Alzheimer’s disease,” said Scott Noggle, PhD, NYSCF, Charles Evans Senior Research Fellow for Alzheimer’s Disease and Director of the New York Stem Cell Foundation (NYSCF)’s laboratory and senior author on the study.
While other groups have recently generated human iPS cells models of Alzheimer’s disease with studies primarily focused on familial Alzheimer’s disease neurons, none of these studies addressed whether there are any differences between Alzheimer’s disease and control neural progenitor cells prior to neuronal differentiation.
Studying neural progenitor cells may also reveal developmental components of familial Alzheimer’s disease, and are more homogenous than the wide variety of neurons currently produced by differentiation protocols, which allows for better comparisons between controls and disease cells.
Dr. Gandy and Dr. Noggle are both members of the Cure Alzheimer’s Stem Cell Consortium that supported this research. The Stem Cell Consortium is an international group of scientists that are working together to directly investigate, for the first time, the brain cells from individuals with the common form of Alzheimer’s disease. Other members of the Consortium include Kevin Eggan, PhD, of Harvard University, Marc Tessier-Lavigne, PhD, of Rockefeller University, Doo Kim, PhD, of Harvard Medical School, and Tamir Ben-Hur, MD, PhD, of Hadassah University.
NYSCF stem cell researcher Andrew Sproul, PhD, a staff scientist at the NYSCF Research Institute is the lead author on this study. Samson Jacob of NYSCF, other NYSCF and ISMMS researchers, scientists at Columbia University, and the James J. Peters VA Medical Center also made significant contributions to this study.
The New York Stem Cell Foundation Research was supported by grants from the Charles Evans Foundation, Alzheimer’s Drug Discovery Foundation, and NY Community Trust. Scott Noggle and Sam Gandy are jointly supported by an NIH grant, and the Cure Alzheimer’s Fund and the other ISMMS research was supported by the American Health Assistance Foundation. The NIH RF1 grant to Drs. Noggle and Gandy is part of an additional $50 million allocated to Alzheimer’s research in fiscal 2012 under the Obama Administration’s “We Can’t Wait” Alzheimer’s initiative. The Columbia University research was supported by two NIH grants.
Abstract of Acta Neuropathologica Communications paper
Background – Induced pluripotent stem cells (iPSCs) derived from patients with neurodegenerative disease generally lack neuropathological confirmation, the gold standard for disease classification and grading of severity. The use of tissue with a definitive neuropathological diagnosis would be an ideal source for iPSCs. The challenge to this approach is that the majority of biobanked brain tissue was not meant for growing live cells, and thus was not frozen in the presence of cryoprotectants such as DMSO.
Results – We report the generation of iPSCs from frozen non-cryoprotected dural tissue stored at -80[degree sign]C for up to 11 years. This autopsy cohort included subjects with Alzheimer’s disease and four other neurodegenerative diseases.
Conclusions – Disease-specific iPSCs can be generated from readily available, archival biobanked tissue. This allows for rapid expansion of generating iPSCs with confirmed pathology as well as allowing access to rare patient variants that have been banked.
Abstract of PLOS ONE paper
Presenilin 1 (PSEN1) encodes the catalytic subunit of γ-secretase, and PSEN1 mutations are the most common cause of early onset familial Alzheimer’s disease (FAD). In order to elucidate pathways downstream of PSEN1, we characterized neural progenitor cells (NPCs) derived from FAD mutant PSEN1 subjects. Thus, we generated induced pluripotent stem cells (iPSCs) from affected and unaffected individuals from two families carrying PSEN1 mutations. PSEN1 mutant fibroblasts, and NPCs produced greater ratios of Aβ42 to Aβ40 relative to their control counterparts, with the elevated ratio even more apparent in PSEN1 NPCs than in fibroblasts. Molecular profiling identified 14 genes differentially-regulated in PSEN1 NPCs relative to control NPCs. Five of these targets showed differential expression in late onset AD/Intermediate AD pathology brains. Therefore, in our PSEN1 iPSC model, we have reconstituted an essential feature in the molecular pathogenesis of FAD, increased generation of Aβ42/40, and have characterized novel expression changes.
(¯`*• Global Source and/or more resources at http://goo.gl/zvSV7 │ www.Future-Observatory.blogspot.com and on LinkeIn Group's "Becoming Aware of the Futures" at http://goo.gl/8qKBbK │ @SciCzar │ Point of Contact: www.linkedin.com/in/AndresAgostini
 Washington
Washington